
review are exceptionally clear and well resolved unfortunately, no spatial scales or, for two of the videos, temporal scales are provided by the authors.
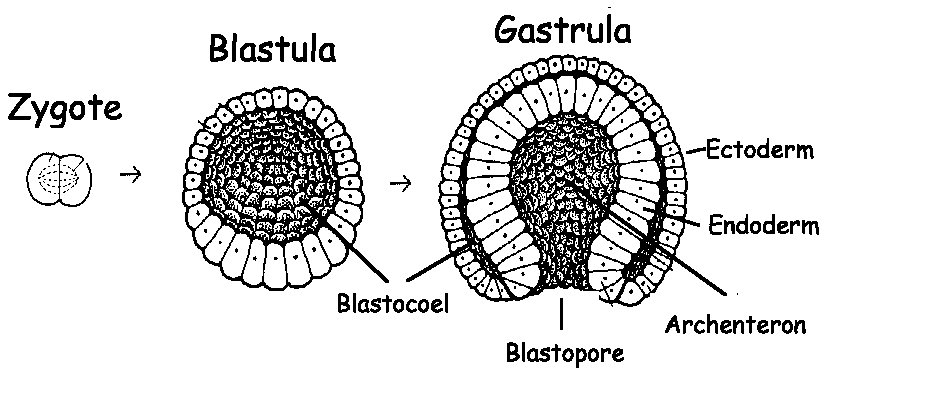
Live sea urchin embryos are also readily studied using phase-contrast or differential interference microscopy (DIC), and the three movies accompanying the McClay et al. Thus, the cellular details of the process are readily apparent, as illustrated in the simple drawings of Figure 1 and reviewed by McClay et al. Historically, undergraduates taking a course in developmental biology have been introduced to gastrulation using sea urchin embryos, which are small, transparent, and easily preserved for whole-mount observation at intermediate levels of magnification. I welcome your comments and questions regarding this review. I review here several videos published as freely accessible supplements to this monograph at along with another recently published video of gastrulation in C. These studies have been extensively documented in a recent monograph that describes the molecular interactions that cause and, in turn, result from gastrulation in embryos from many different animal taxa (Stern, 2004). With the widespread use of fluorescence microscopy and such fluorescent probes as green fluorescent protein (GFP), a renaissance in the study of cellular movements during gastrulation in living embryos began in the latter part of the twentieth century. Then they could formulate predictions of how those features might change during gastrulation in other organisms. Students studying the underlying cellular details of gastrulation might be encouraged to first examine the process in the development of one organism and analyze the various features of cellular movement. New biochemical influences and physical interactions accompany these changes in cellular neighborhoods. During gastrulation, many if not all of the newly cleaved cells dramatically change their location within the embryo in a variety of different ways, sometimes individually and sometimes as contiguous sheets of cells. Indeed, as one developmental biologist has remarked, ‘‘It is not birth, marriage, or death, but gastrulation which is truly the most important time in your life’’ (Wolpert, 1991). The process is all the more impressive for the relative rapidity of its passage and for its pivotal role in bringing about subsequent morphogenetic events in the life history of an animal. This transforma- tion is so universal among animal embryos that developmental biologists refer to the process with a single term: ‘‘gastrulation.’’ Dramatic changes in embryonic complexity accompany gastrulation. In contrast, it is not obvious how the embryonic mass of cells resulting from cleavage becomes transformed into a multilayered, functionally complex, and, in many instances, bilaterally symmetrical embryo.
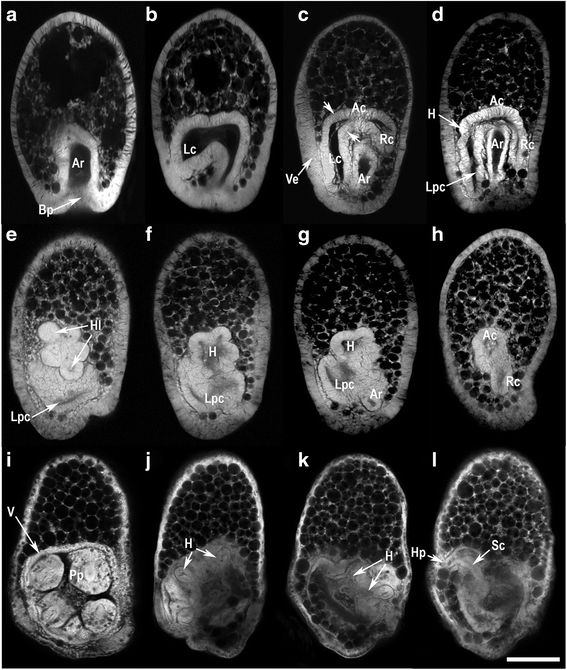
the most naive student readily understands that following fertilization, a single-celled egg must undergo multiple rounds of cell division to become a multicellular organism. Except for the basal character of expression in endoderm and endomesoderm, these transcription factors are clearly utilized differently in chordates. Hphnf3 may contribute to specification of embryonic cells as archenteron, and the role of HpTa may be directed towards specification of mesodermal founder cells. After the onset of gastrulation, however, Hphnf3 is expressed in the posterior part of the archenteron, whereas the Brachyury orthologue, HpTa, is expressed in the secondary mesenchyme founder cells, which occupy the anterior tip of archenteron. The forkhead and Brachyury genes begin to be expressed almost simultaneously in sea urchin embryos, in the vegetal plate at the late blastula stage. At the prism-embryo stage, the entire archenteron stained intensely then, at pluteus stage, the larva staining decreased in intensity. During gastrulation, intense staining was evident in the cells surrounding the blastopore, whereas weak staining was detected in the invaginating archenteron. In situ hybridization signals were found in cells of the vegetal plate of the swimming blastula. An Hphnf3 transcript of 3.0 kb was first detected at the swimming blastula stage, accumulated maximally at the gastrula and prism-embryo stages, and decreased at the pluteus-larva stage. Here we report the pattern of expression of a sea urchin orthologue of forkhead, Hphnf3 which is present as a single copy per haploid genome. To gain insight into developmental mechanisms important in the origin and early evolution of chordates, we investigated sea urchin homologues of chordate genes that are implicated in notochord formation, viz. They lack a notochord or any structures obviously homologous with it. Echinoderms are the sister group of the chordates and hemichordates within the deuterostomes.
